can be reached 24/7
Safe, proven-effective, non-medication approach to the treatment of depression and OCD.
The exact cause of depression is not known. Leading scientific theory is that depression is caused by an imbalance of the brain’s neurotransmitters, chemical messengers that send signals between brain cells. While antidepressant medications may be effective for many patients, they do not work for everybody. NeuroStar® Advanced TMS Therapy is a safe, proven-effective, non-medication approach to the treatment of depression. Additionally, TMS Therapy is now approved for the treatment of Obsessive Compulsive Disorder (OCD) in Adults. Download OCD Brochure
NeuroStar TMS is now approved for treatment of Adolescent Depression (age15+). TMS is a non-invasive, non-medication treatment that uses focused magnetic pulses to “wake up” areas of the brain and help your brain work the way it should. Download Adolescent Depression Brochure.
For inquiries and information, please contact the TMS program by calling 603.434.1577, option 3. For more information visit, TMSatCLM.org
Find out if TMS is right for you.
*Screening tool results will be shared with NeuroStar, who works in partnership with Center for Life Management to provide safe, proven-effective, non-medication approach to the treatment of depression through TMS treatment. By clicking Take The Quiz you are agreeing to these terms.
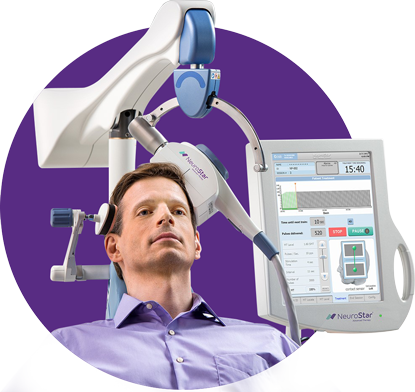
What Is NeuroStar Advanced TMS Therapy?
Transcranial Magnetic Stimulation (TMS) is a breakthrough non-medication treatment for major depression in adults and adolescents age 15+. NeuroStar TMS is safe and proven effective for those who have not responded to treatment with antidepressant medications.
NeuroStar TMS offers new hope for people with depression. Signs of depression include:
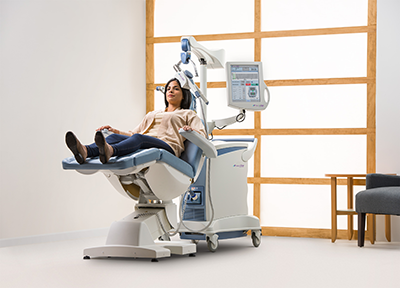
How Does NeuroStar TMS Therapy Work?
NeuroStar TMS is done on an outpatient basis by prescription only and under the supervision of a psychiatrist. During treatment, the patient is awake and seated in a comfortable chair, much like a dentist’s chair. A small, cushioned magnetic coil will be positioned lightly on your head.
Does Receiving TMS Hurt? What Does It Feel Like?
NeuroStar delivers focused magnetic stimulation directly to the correct location that regulates mood in the brain.
You’ll hear a clicking sound and may feel a tapping, tingling or warm sensation on your head where the cushioned coil is positioned. For the vast majority of people, TMS treatment does not hurt. Ultimately, everyone’s experience is different. If you feel any discomfort, be sure to tell your treatment team.
What Happens After TMS Treatment? Can I Return to My Normal Activities?
Depending on your doctor’s recommendation, each treatment takes between 19 and 37 minutes.
You can resume normal activities immediately. Because there are no effects on alertness or memory, you can drive yourself to and from treatment sessions.
In-office treatment with NeuroStar TMS Therapy takes 19-37 minutes and is administered 4-5 days a week for approximately 4-6 weeks. For best results, it’s important to complete the full number of treatment sessions prescribed by your doctor.
What are the Side Effects?
NeuroStar TMS is a non-medication treatment and has none of the systemic side effects commonly associated with antidepressant medication. TMS treatment is generally tolerated by patients with minimal side effects, the most common being mild scalp discomfort.
NeuroStar TMS is an FDA-approved device and TMS treatment has been proven safe and effective in over 20 years of research on the treatment of depression.
Ready to make an appointment?
If you think NeuroStar TMS might be right for you, contact CLM’S TMS Program and find out more.
Please call our CLM TMS Program
Call (603) 434-1577 Option 3